Regrowing cartilage.
Restoring mobility.
Redefining OA therapy.
Stemora is developing the first one-step, disease-modifying therapy designed to regenerate native hyaline cartilage — activating the body's own skeletal stem cells to repair the tissue every other osteoarthritis treatment only masks.
OA isn't a pain problem.
It's a tissue-loss problem.
Cartilage doesn't heal on its own. The current standard of care — NSAIDs, steroids, hyaluronic acid, microfracture, MACI, joint replacement — manages symptoms or mechanically replaces the joint. None regenerate the native hyaline cartilage that joints need.

From the first sharp injury to the inevitable replacement, patients deserve biology, not workarounds.
One procedure.
Native cartilage. Regenerated.
Stemora combines microfracture surgery with BMP2 plus a VEGF inhibitor (cabozantinib) to activate the patient's own resident skeletal stem cells — and direct them, in a single outpatient procedure, toward true hyaline cartilage. No cell harvest. No synthetic implants. No repeat visits.
Activate the body's own stem cells
Microfracture creates micro-channels into the subchondral bone, drawing the patient's resident skeletal stem cells (SSCs) into the defect site — the mechanical trigger.
Guide them down the cartilage lineage
BMP2 pushes SSCs toward chondrogenesis. A VEGF inhibitor blocks vascular invasion and the default drift toward bone or stroma. Together, they direct fate to cartilage.
Regrow native hyaline cartilage
The result: durable, localized regeneration of Type II collagen and aggrecan-rich tissue — the same biological cartilage the joint was built to use, not fibrocartilage scar.
For everyone whose joints
keep them in motion.
Stemora is built for athletes, active patients, weekend warriors, military operators, and anyone whose life depends on the joint working. Cartilage loss takes them out of the game. Regeneration brings them back.
The only therapy designed
to regenerate native hyaline cartilage in one procedure.
Microfracture produces fibrocartilage. MACI requires multi-stage cell harvest. Implants and prosthetics deliver mechanical workarounds — not biology. Stemora is engineered to deliver what the field has been missing.
| Feature | Stemora | Microfracture | MACI / Cell Therapy | Implants & Replacement |
|---|---|---|---|---|
| Tissue regenerated | Native hyaline cartilage | Fibrocartilage (Type I) | Hyaline-like / mixed | None — mechanical only |
| Procedure | One-step outpatient | Single-stage | Multi-stage + cell harvest | Highly invasive surgery |
| Durability | Designed for biologic, long-lasting | Declines in 2–5 yrs | Variable | Non-biologic; revision risk |
| Recovery | Shorter, weight-bearing compatible | Restricted | Long, repeat visits | Long rehabilitation |
| Cost & scalability | Moderate · fits existing OR workflow | Low / high | High / cell-limited | Very high / low |
| Regulatory path | FDA 505(b)(2) | Approved procedure | BLA | 510(k) / PMA |
Cartilage regeneration
validated across three preclinical models.
Stemora's platform is grounded in peer-reviewed research from Stanford Medicine. The mechanism has been demonstrated in mouse OA, human cartilage xenograft, and minipig large-animal models — each showing hyaline cartilage regeneration markers, not scar tissue.
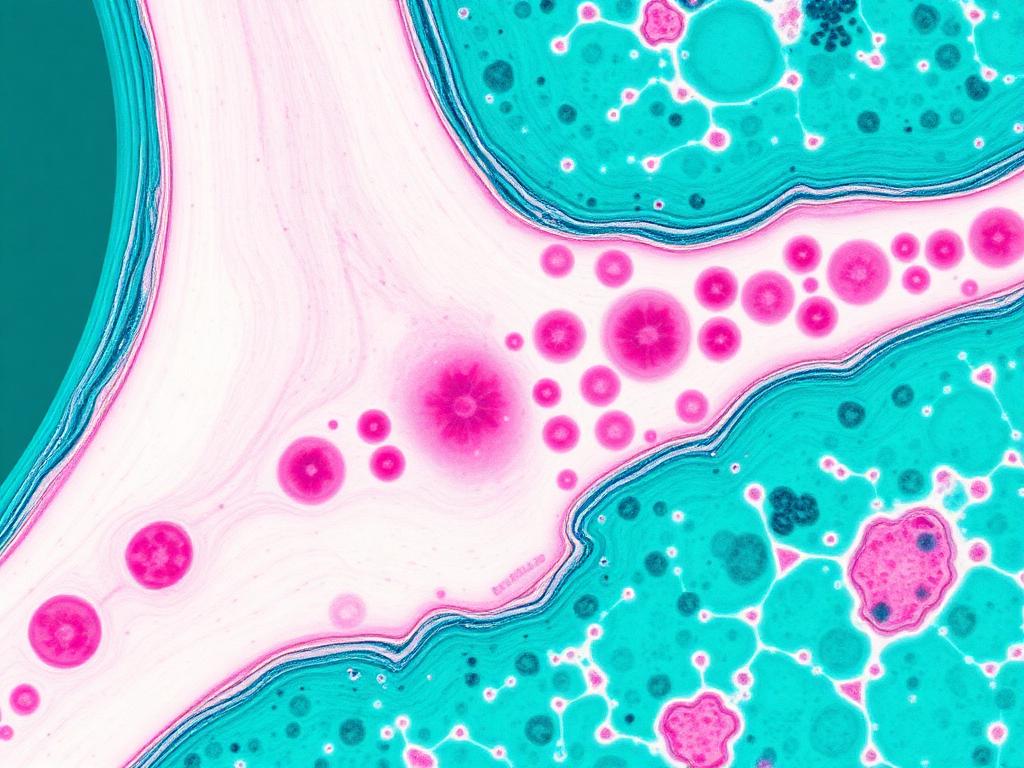 Safranin-O · cartilage histology
Safranin-O · cartilage histologyMouse OA model
Microfracture combined with BMP2 + VEGF blockade regrew cartilage at defect sites with ACAN and Type II collagen markers — and minimal fibrocartilage or hypertrophic bone signal vs. PBS control.
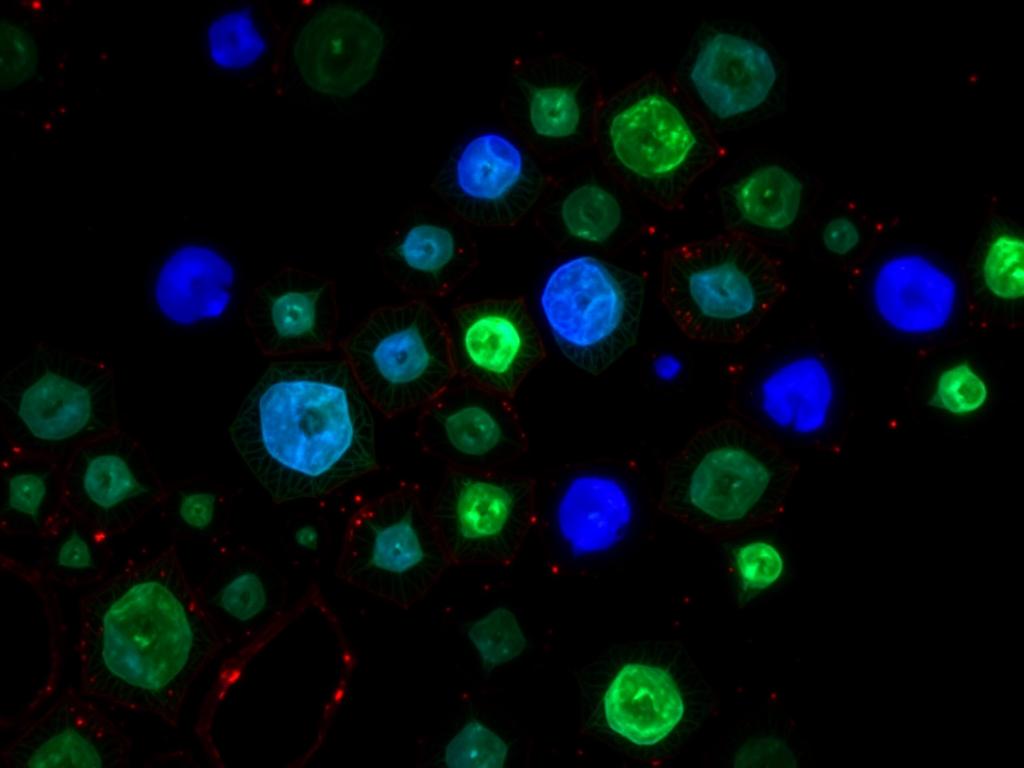 Immunofluorescence · human SSC
Immunofluorescence · human SSCHuman cartilage xenograft
Human tissue in NSG mouse xenografts validated that human skeletal stem cells respond to the same molecular cues — supporting translation from mouse biology to clinic.
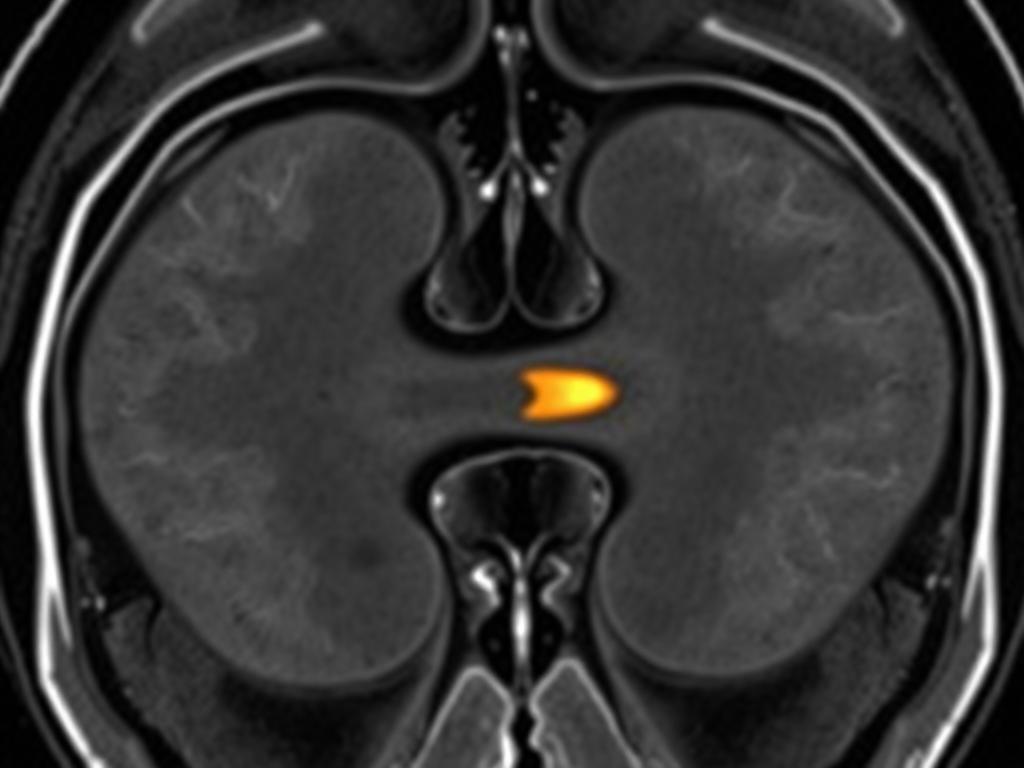 MRI · minipig joint scale
MRI · minipig joint scaleMinipig large-animal
Gross, histological pentachrome, and MRI-confirmed regeneration of hyaline cartilage in a joint-scale model — the closest preclinical analog to human knee anatomy.
Multi-billion-dollar opportunity
with a clear commercial wedge.
Stemora enters through focal cartilage defects compatible with microfracture — a sizable, well-defined population that maps directly onto existing orthopedic surgical workflows — then expands into the broader knee and OA market.
Milestone-driven plan,
built for value inflection.
A staged $60–65M investment over six years is projected to yield 5.4×–8.3× returns (IRR ~35–46%) — supported by capital efficiency, exclusive IP, and an accelerated regulatory strategy through FDA 505(b)(2).
$350M–$500M strategic acquisition
Identified strategic acquirers: Medtronic, Stryker, DePuy Synthes — each actively pursuing biologic differentiation in joint preservation. Series C or IPO are alternative paths.
Built by the scientists
who originated the work.
The Stemora team combines the Stanford-published research team behind the mechanism with operators experienced in biotech development, FDA strategy, and orthopedic surgery.





Questions investors and
partners ask first.
Help us build the first
regenerative therapy for OA.
Stemora's Pre-Seed SAFE round is open with a $1M target and Q1 2026 close. Series A and B targets, capital plan, and projected returns are available under NDA.



